How GOOD Paint Removers Work on multiple layers
Paint removers work by penetrating the coating, breaking chemical bonds, and softening or swelling the paint film so it can be scraped off. They do this in two main ways:
1. Solvent‑Based Paint Removers
These are the fastest and most common. They work by penetrating the paint, swelling it, and breaking the bond between layers and the surface.
We love Welsh Fern Oil
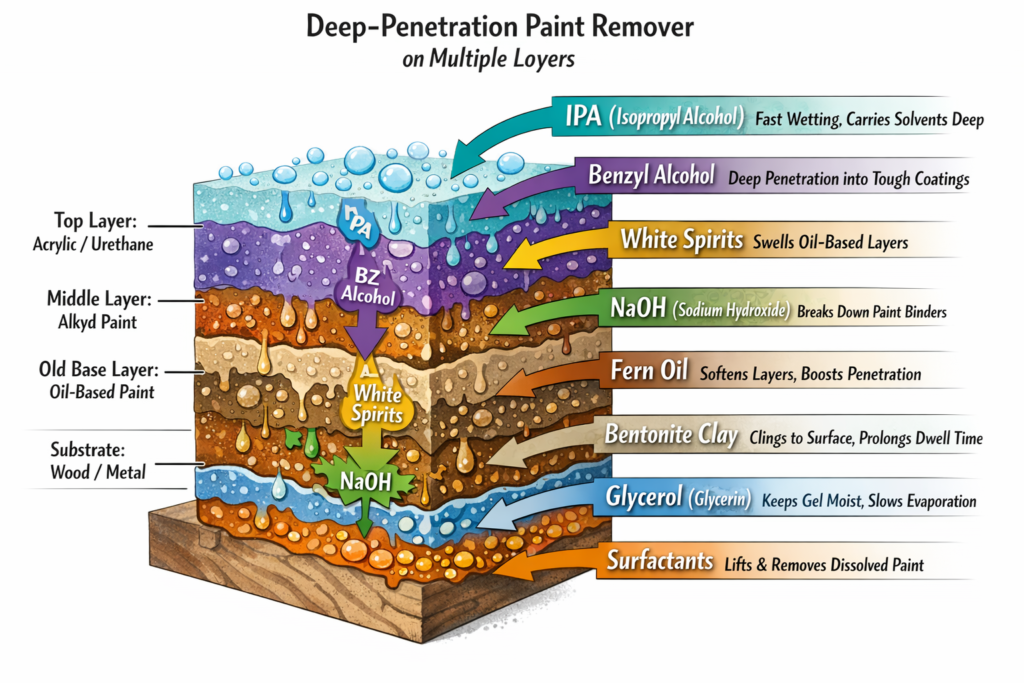
How They Remove Multiple Layers
Solvents soak through the top coat, then continue downward.
As each layer swells, it blisters and lifts, exposing the next layer.
Thick, old paint may require reapplication because solvents evaporate or get consumed as they penetrate deeper.
🔍 Key Ingredients & What They Do
Ingredient Function Notes
Methylene chloride (dichloromethane) Rapid penetration; swells paint films Historically most effective; now restricted due to safety risks
Benzyl alcohol Slower but safer solvent; penetrates deeply Common in modern “safer” strippers
N‑Methyl‑2‑pyrrolidone (NMP) Dissolves or lifts paint polymers Effective but under regulatory scrutiny
Dimethyl adipate / dibasic esters Swell paint slowly; low volatility Used in “green” formulations
Acetone / MEK Fast-evaporating solvents for lacquers Limited contact time; flammable
🧬 How Solvents Work Chemically
They penetrate the paint film, opening pathways for water/alcohols inside the formula.
The paint swells, breaking intermolecular forces.
The coating pops loose from the substrate.
2. Caustic (Alkaline) Paint Removers
These use strong bases to chemically break down paint.
How They Remove Multiple Layers
Caustics react with oils and resins in paint (saponification).
They break down the polymer structure, turning paint into a soft, jelly-like mass.
They continue reacting as long as the chemical stays wet, making them excellent for very thick, old, multi‑layered paint.
Key Ingredients & What They Do
Ingredient Function Notes
Sodium hydroxide (NaOH) Breaks chemical bonds; saponifies oils Highly effective on old oil-based paints; corrosive
Potassium hydroxide (KOH) Similar to NaOH; strong base Used in some heavy-duty strippers
🧬 How Caustics Work Chemically
They attack the paint’s chemical structure, not just the bond.
They break long polymer chains into smaller, water‑soluble fragments.
The paint softens and releases from the surface.
Layer‑by‑Layer Removal: What Actually Happens
Whether solvent or caustic, the process looks like this:
Top layer softens first
Solvents swell it; caustics break it down.
Blistering occurs
The paint bubbles as layers detach from each other and the substrate.
Underlying layers begin to lift
Solvents penetrate downward.
Caustics continue reacting with deeper layers.
Scraping removes multiple layers at once
Thick paint often comes off in sheets.
Reapply for stubborn deep layers
Especially with solvent-based gels, which may dry before reaching the bottom layers.
Why Some Products Work Better on Many Layers
Best for thick, old, multi‑layer paint:
Caustic strippers (NaOH-based)
They keep reacting as long as they stay wet.
Excellent for architectural restoration.
Best for modern coatings or mixed paint types:
Solvent-based strippers (benzyl alcohol, esters)
More universal compatibility.
Safer than older methylene chloride formulas.
Safety Notes (Important)
Solvents can be toxic or flammable.
Caustics can cause severe burns.
